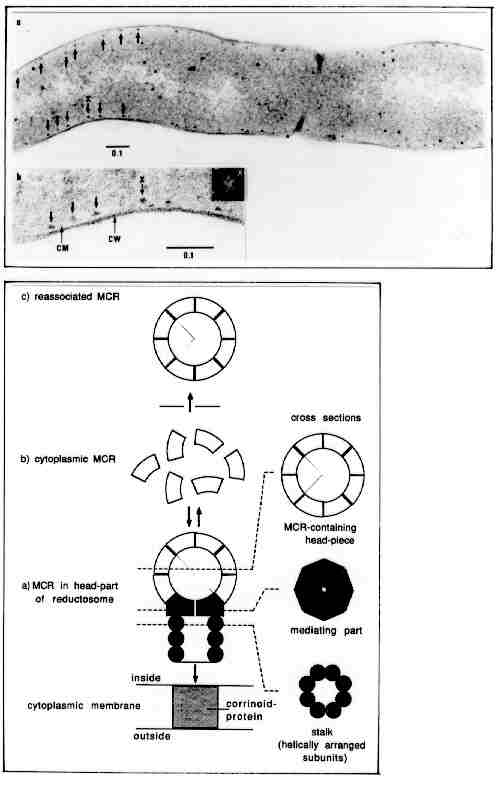

Top figure:
a, b, ultrathin section of Methanobacterium thermoautotrophicum.
The arrows point to particulate entities seen close to the cytoplasmic
membrane. They are interpreted to the methanoreductosomes attached to the
cytoplasmic membrane by a stalk (s. below). One of them (x) exhibits clear
symmetric substructures. Dimensions are given in µm.
Bottom figure:
Diagrammatic view of the enzyme complex consisting of a head part (containing
several copies of the enzyme methyl-CoM reductase; MCR), a mediating part,
and a stalk which links the particle to a component of the system in the
cytoplasmic membrane (a).
b, isolated enzyme methyl-CoM reductase, which was used for reconstitution
experiments and which is assumed to also occur in the cell cytoplasm.
c, result of reconstitution experiments: MCR particles assemble to
form hollow spheres (shells) similar in size to head parts of methanoreductosomes
isolated from the cells; however, the reconstituted head parts contain
MCR only and they did not show any polarity, as is the case for in vivo
head parts. The interaction of the end of the stalk with a membrane-integrated
corrinoid protein (methyltransferase) has not been shown. (Upper figure:
original micrograph taken by F. Mayer and M. Hoppert; bottom figure: from
Mayer, F., 1993, ASM News 59, 346-350; additional references therein).