HPr kinase of
Mycoplasma pneumoniae - a key regulatory
protein that mirrors the parasitic lifestyle of Mycoplasma
 Mycoplasmas are pathogens of mammals that cause respiratory and
urogenital diseases. They belong to the low GC Gram-positive bacteria even
though they lack a cell wall. Their parasitic life is reflected by the fact
that they contain the smallest genomes of any self-replicating organisms
known so far. Over the past years, mycoplasmas have attracted considerable
and growing interest owing to three considerations: (i)
to understand the biology of an important pathogen; (ii) to identify the
minimum genetic equipment necessary for independent cellular life and to
construct such minimal genomes and, (iii) to develop and refine the
methodology in the post-genomic era such as proteomics and transcriptomics
Among the very few regulatory proteins encoded by Mycoplasma
pneumoniae is HPr kinase/phosphatase (HPrK/P), the
key regulator of carbon metabolism in low GC Gram-positive bacteria. The
activity of HPr kinase is modulated by the metabolic state of the cell. In
contrast to HPrK/P of B. subtilis, which is by default a phosphatase and
needs high ATP concentrations for kinase activity, the M. pneumoniae derived
enzyme exhibits kinase activity at very low ATP concentrations and depends on
inorganic phosphate for phosphatase activity. This inverted control of
enzymatic activity may result from the adaptation to very different ecological
niches. To understand the functionaliziation of HPrK/P, we are going to
investigate its crystal structure and to probe its interaction with HPr and
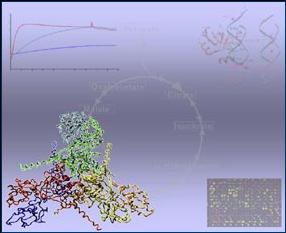
different metabolites using biochemical and spectroscopical methods. Figures:
The centrol role of HPr kinase/phosphatase in the regulation of carbon
metabolism Crystals of HPrK/P Mycoplasmas are pathogens of mammals that cause respiratory and
urogenital diseases. They belong to the low GC Gram-positive bacteria even
though they lack a cell wall. Their parasitic life is reflected by the fact
that they contain the smallest genomes of any self-replicating organisms
known so far. Over the past years, mycoplasmas have attracted considerable
and growing interest owing to three considerations: (i)
to understand the biology of an important pathogen; (ii) to identify the
minimum genetic equipment necessary for independent cellular life and to
construct such minimal genomes and, (iii) to develop and refine the
methodology in the post-genomic era such as proteomics and transcriptomics
Among the very few regulatory proteins encoded by Mycoplasma
pneumoniae is HPr kinase/phosphatase (HPrK/P), the
key regulator of carbon metabolism in low GC Gram-positive bacteria. The
activity of HPr kinase is modulated by the metabolic state of the cell. In
contrast to HPrK/P of B. subtilis, which is by default a phosphatase and
needs high ATP concentrations for kinase activity, the M. pneumoniae derived
enzyme exhibits kinase activity at very low ATP concentrations and depends on
inorganic phosphate for phosphatase activity. This inverted control of
enzymatic activity may result from the adaptation to very different ecological
niches. To understand the functionaliziation of HPrK/P, we are going to
investigate its crystal structure and to probe its interaction with HPr and
different metabolites using biochemical and spectroscopical methods. Figures:
The centrol role of HPr kinase/phosphatase in the regulation of carbon
metabolism Crystals of HPrK/P
Novel tools for
the genetic analysis of Mycoplasma pneumoniae
Key references
Steinhauer, K., Jepp, K., Hillen, W. &
Stülke, J. (2002) A novel mode of
control of Mycoplasma pneumoniae
HPr kinase/phosphatase activity reflects its
parasitic life style. Microbiology 148: 3277-3284.
Allen, G. S., Steinhauer, K., Hillen, W., Stülke, J. & Brennan, R. G. (2003) Crystal
structure of HPr kinase/phosphatase from Mycoplasma pneumoniae.
J. Mol. Biol. 326: 1203-1217.
Merzbacher, M., Detsch, C., Hillen, W. &
Stülke, J. (2004) Mycoplasma pneumoniae
HPr kinase/phosphorylase: assigning functional
roles to the P-loop and the HPrK/P signature
sequence motif. Eur. J. Biochem.
271: 367-374.
Halbedel, S., Hames, C. & Stülke, J. (2004) In vivo activity of enzymatic and regulatory components of the phosphoenolpyruvate:sugar phosphotransferase system in Mycoplasma pneumoniae. J. Bacteriol.
186: 7936-7943.
|

